- Home
- About the CAP
- Annual Report 2025
-
-
In 2025, the College of American Pathologists (CAP) delivered measurable results for pathologists, laboratories, and the patients they serve. We strengthened education and leadership development, advanced laboratory quality and accreditation, expanded innovation in practice, and secured policy wins that protect patient access to high-quality testing while maintaining strong financial stewardship to support our mission for years to come. Our work this year reflects a clear priority: Ensuring pathologists have the standards, tools, and voice to lead diagnostic medicine.
Membership remains the foundation of the CAP. In 2025, we exceeded 20,000 members for the first time in our history. Through new initiatives for members like the OnDemand education platform and AI Studio, we’re equipping pathologists with greater access to the latest innovations and best practices. Importantly, our programs provide the skills, connections, and tools needed to lead with confidence and advance the profession of pathology and laboratory medicine.
Our Laboratory Quality Solutions drive excellence in patient care through accreditation and proficiency testing. We accredit more than 8,400 laboratories worldwide and now serve as the official accreditor for US Department of Veterans Affairs laboratories. We also introduced new—and some industry-first—proficiency testing/external quality assessment (PT/EQA) programs and advanced evidence-based guidelines to ensure accuracy, consistency, and excellence in diagnostic care.
Our advocacy further amplified the voice of pathology nationwide. CAP members mobilized on Capitol Hill and across the US, securing meaningful federal and state policy victories that preserve access to quality testing and support the long-term viability of our profession.
The CAP led with purpose—embracing innovation, strengthening our community, and positioning pathology to meet the challenges and opportunities of a rapidly evolving health care landscape.
Qihui "Jim" Zhai, MD, FCAP
CAP PresidentAkshay Patel
CAP Interim CEO
CAP Board of Governors

The CAP Board of Governors, from left to right, sitting: Guillermo G. Martinez-Torres, MD, FCAP; M.E. (Doc) de Baca, MD, FCAP; Rebecca L. Johnson, MD, FCAP; Alfred Wray Campbell, MD, MBA, FCAP (Secretary-Treasurer); Qihui "Jim" Zhai, MD, FCAP (President); Kalisha Ashara Hill, MD, MBA, FCAP (President-Elect); Akshay Patel (Interim CEO); Marilyn M. Bui, MD, PhD, FCAP (House of Delegates Vice Speaker); and Bharati Suketu Jhaveri, MD, FCAP (CAP Foundation President). From left to right, standing: Raj C. Dash MD, FCAP; Cullen M. Lilley, MD, MS, MA (Residents Forum Executive Committee Chair); Aaron C. Han, MD, PhD, FCAP; Bradley S. Karon, MD, PhD, FCAP; Donald S. Karcher, MD, FCAP (Immediate Past President); A. Joe Saad, MD, CPE, FCAP; Earle S. Collum, MD, FCAP; Diana Marcella Cardona, MD, MBA, FCAP; Joel Todd Moncur, MD, PhD, MS, FCAP; Sang Wu, MD, FCAP (House of Delegates Speaker); Aaron Auerbach, MD, MPH, FCAP.


In 2025, the CAP provided its more than 20,000 members with expanded access to education, leadership development opportunities, peer connection, and emerging technologies to help pathologists strengthen their voices and navigate a rapidly evolving profession.
Education remained central to supporting members across all career stages and practice settings. The CAP delivered 80 live and virtual education courses in 2025, awarding 431,093 continuing medical education (CME) credits.
In addition to live programming, the CAP launched CAP On Demand, a member-exclusive streaming education platform offering 24/7 on-demand access to presentations across subspecialty pathology, laboratory management, and leadership topics from past CAP annual meetings. Since its launch, 218 video/audio clips were uploaded, and 401 members created accounts.
Leadership development remained a strategic priority through the Engaged Leadership Academy (ELA), empowering pathologists at all career stages. The ELA offered immersive training in communication, presentation skills, conflict resolution, and leadership strategy. Small group coaching and mentorship helped members develop practical tools for real-world challenges.
In addition to its year-round leadership development efforts, the CAP introduced a new full-day leadership program held in conjunction with the 2025 House of Delegates & Pathologists Leadership Summit. The workshop was highly rated by participants, with 98% satisfied or very satisfied with their overall Summit experience and 100% rating the leadership workshop’s value as excellent or good.
The CAP strengthened peer connection through the launch of PathTogether, a secure, members-only online community that fosters discussion, knowledge sharing, and professional support. The platform enables members to connect across subspecialties, practice settings, and career stages. Within the first four weeks, 750 members from 32 countries were engaging on the platform.
Recognizing the growing role of technology in pathology, the CAP introduced the AI Studio, a secure, web-based environment that allows members to explore emerging AI tools. By interacting with AI models, members can evaluate potential applications, gain practical insight, and build confidence in how these tools may enhance their work. Since its launch, more than 400 members have used the AI Studio, completing nearly 2,000 sessions across 39 models.
Through these initiatives, the CAP continued investing in its members by delivering education, leadership development, meaningful connections, and practical tools that support pathologists at every stage of their careers.
80
live and virtual education courses delivered
431,093
CME credits awarded
750
members connected through PathTogether



The CAP continues to set the gold standard in laboratory accreditation and proficiency testing/external quality assessment (PT/EQA) programs. In 2025, the CAP became the official accreditor for US Department of Veterans Affairs laboratories, further reinforcing its leadership in laboratory quality and oversight. The CAP now accredits more than 8,400 laboratories across 60 countries, with over 750 laboratories outside of the US and Canada.
While offering more than 700 PT/EQA programs across 16 disciplines, the CAP introduced 21 new programs in 2025. More than 23,300 laboratories rely on CAP PT/EQA programs to ensure consistent quality, accuracy, and reliability in testing.
Among the 21 new PT/EQA offerings released in 2025 were several industry-first programs, including the first global HIV-1/HIV-2 molecular detection and differentiation program and the first US-based PT program for detecting and accurately identifying the H5N1 strain of avian influenza. We’re also the first US-based provider to offer a standalone program for Dengue virus serology.
Additionally, the CAP advanced patient care through its evidence-based guidelines, releasing an updated guideline on human papillomavirus (HPV) testing in head and neck carcinomas, as well as a new guideline to standardize amyloidosis testing.
23,300
Laboratories using CAP PT/EQA programs
8,400
CAP-accredited laboratories worldwide
21
new PT/EQA programs launched


All across the country, CAP members stepped forward in 2025 to shape the policies that affect their profession and their patients. By engaging lawmakers, taking collective action, and supporting targeted advocacy efforts, the CAP secured key wins that strengthened pathology and preserved access to high-quality testing.
Pathologists engaged lawmakers through both virtual and in-person Hill Days, with 175 total advocates participating in 225 meetings on Capitol Hill. CAP’s PathNet program mobilized 2,202 advocates who sent 19,165 action alert messages across 11 campaigns, reaching every US senator and 391 members of the House of Representatives.
A major federal victory came when a US District Court judge struck down the FDA's laboratory-developed test (LDT) regulation, agreeing with CAP and fellow plaintiffs that the rule exceeded the agency’s authority. The decision protected patient access to timely diagnostic testing throughout the country.
CAP advocacy also delivered progress on payment policy. Congress included a 2.5% Medicare physician payment increase for 2026 in the "One Big Beautiful Bill Act," offering temporary relief while the CAP continues to press for long-term reform of the Medicare payment system. Laboratories also received a reprieve from the PAMA cuts of up to 15% when Congress acted to avert a government shutdown. And, after years of advocacy, we overturned a restrictive Medicare policy, securing coverage for special stains in key cancer pathology services.
Political engagement through PathPAC remained strong, with pathologists contributing more than $215,000. The PAC supported 102 candidates and candidate-affiliated PACs with $214,000, helping ensure pathologists’ voices are heard in key policy discussions.
At the state level, CAP legislative language was included in five amended state laws and in new American Medical Association policies that will help pathologists and patients throughout the country.
19,165
action alert messages to Congress
2,202
CAP member advocates
$215,000
PathPAC contributions


The CAP delivered a solid financial performance during the year, navigating revenue headwinds through disciplined financial management and select one-time business factors in support of patients, members, and customers. Revenue was close to the Board of Governors–approved target, as international shipping restrictions delayed proficiency testing deliveries, timing-related impacts of publication schedules, and changes to learning offerings were largely offset by growth in accreditation from new laboratories, the recapture of the Veterans Administration contract, and stronger than expected annual meeting sponsorship and periodicals advertising. Expenses were significantly below budget, reflecting effective cost management, including workforce optimization, lower benefit utilization, and favorable timing of proficiency testing shipments. In addition, there was an unanticipated delay in outside services spending related to the newly installed enterprise resource planning system.
As a result, the CAP exceeded its operational financial targets (as measured by earnings before interest, depreciation, and amortization, minus capital expenditures (EBIDA less CapEx)) delivering positive results of $11.9 million, which is $14.9 million better than budget—strengthening the organization’s ability to invest in its mission and future priorities.
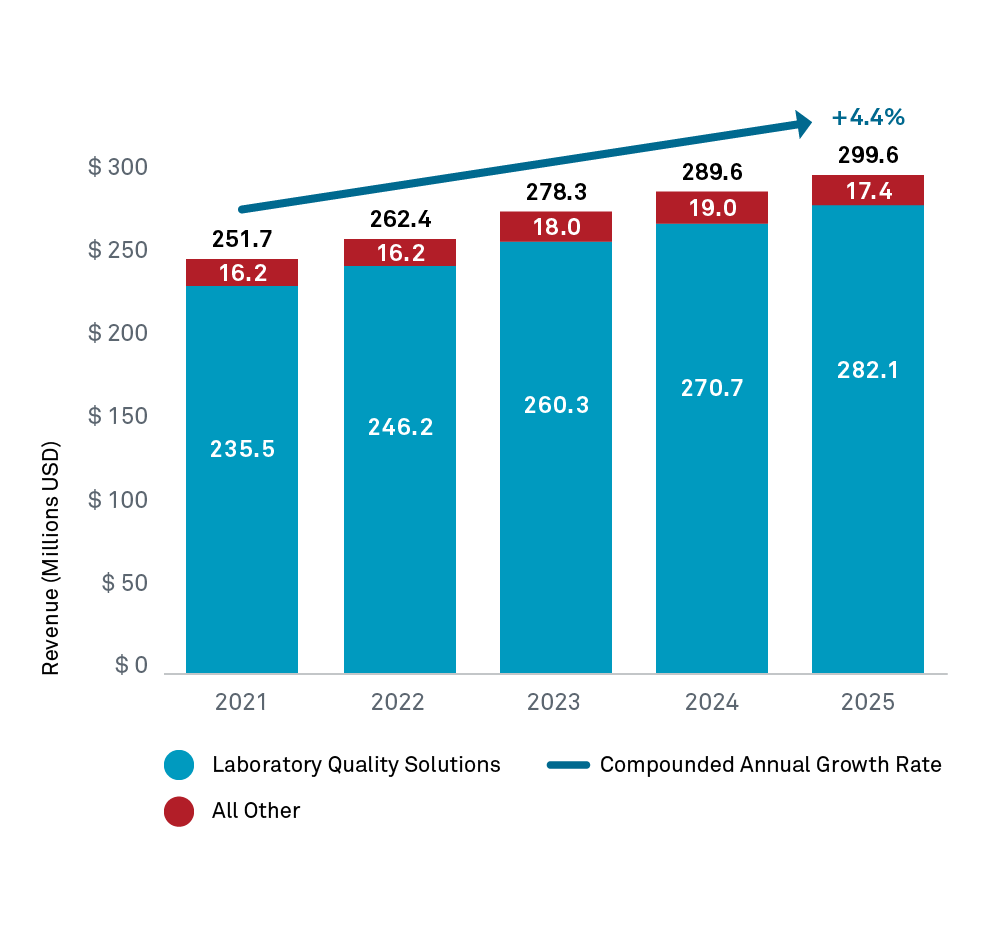
Program Revenue
| Revenue Category | 2025 Amount (millions USD) | % of Total Operating Revenue |
|---|---|---|
| Program Revenue(1) | 215.8 | 72.1% |
| Laboratory Accreditation Fees | 66.8 | 22.3% |
| Standards & Guidelines Revenue | 4.7 | 1.6% |
| Periodical and Published Materials | 4.4 | 1.5% |
| Membership Dues | 3.8 | 1.3% |
| Learning Revenue | 2.4 | 0.8% |
| Other Revenue | 1.6 | 0.5% |
| Total Operating Revenues | 299.6 | 100.0% |
| Investment Net Earnings / (Loss) | 20.8 | 6.9% |
| Total Revenues | 320.3 | 106.9% |
(1) Program revenue includes proficiency testing and Pathologists Quality Registry revenue.
Expenses
| Expense Category | 2025 Amount (millions USD) | % of Total Expenses |
|---|---|---|
| Personnel and Benefits(2) | 105.9 | 37.5% |
| Cost of Materials and On-site Inspections | 103.4 | 36.7% |
| Outside Services(2) | 31.5 | 11.2% |
| Rental and Maintenance | 17.0 | 6.0% |
| Depreciation and Amortization | 7.4 | 2.6% |
| Council and Committee Expenses | 5.3 | 1.9% |
| Travel | 4.1 | 1.4% |
| Office Expenses | 3.1 | 1.1% |
| General and Administrative | 1.8 | 0.7% |
| Other Expenses | 2.6 | 0.9% |
| Total Expenses | 282.2 | 100.0% |
(2) Amounts for personnel and benefits and outside services have been reduced by the capitalized expenses of $2.4 million and $9.4 million, respectively.
Assets
| Asset Class | Balance as of December 31, 2025 (millions USD) |
|---|---|
| Cash and Cash Equivalents | 13.6 |
| Accounts Receivable | 157.4 |
| Other Current Assets | 35.5 |
| Land, Buildings, and Equipment | 48.8 |
| Net of Accumulated Depreciation | |
| Investments and Other Long-term Assets | 168.6 |
| Total Assets | 423.9 |
| Less: Total Liabilities | 253.7 |
| Net Assets | 170.3 |







