- Home
- Laboratory Improvement
- Accreditation
- Laboratory Accreditation Program
The CAP's Laboratory Accreditation Program is unlike any other. It’s an engaging, dynamic, collaborative experience that fosters continuous improvement. Top-ranked hospitals prefer our program. Why? We help them manage changes in laboratory medicine and technology, as well as the regulatory landscape.
The Centers for Medicare & Medicaid Services (CMS) granted the CAP Laboratory Accreditation Program deeming authority, which allows for a CAP inspection in lieu of a CMS inspection. In addition, the CAP retains deemed status with the Joint Commission, the United Network for Organ Sharing, the National Marrow Donor Program, the Foundation for the Accreditation of Cellular Therapies, and many US state agencies.
Discipline-Specific Checklists Simplify Compliance
Annual checklist updates incorporate the latest best practices. Our checklists, developed with input from more than 500 pathologists, provide a clear roadmap for achieving accreditation and running a high-quality laboratory.
The Power of Peer Inspections
The CAP’s unique peer-inspection model benefits both the laboratories being inspected and those providing the inspection teams. Our inspection teams are made up of trained, practicing laboratory professionals who understand the workflows and challenges you face—because they face them every day. In addition, only the CAP offers specialty inspectors for key, high-complexity disciplines.
Program Benefits
CAP laboratory accreditation helps laboratories:
- Maintain accuracy of test results and ensure accurate patient diagnosis.
- Meet required standards from CLIA, FDA, and OSHA. CAP requirements commonly exceed the standards, bolstering patient care and safety.
- Manage rapidly evolving changes in laboratory medicine and technology.
- Exchange ideas and best practices among pathology and laboratory medicine peers.
- Offer professional development and learning opportunities to their laboratory staff.
Program Details
- On-site inspections occur every two years, using the CAP Accreditation Checklists to assess compliance with program requirements.
- Participating laboratories can access the checklists through e-LAB Solutions Suite, while non-accredited laboratories may purchase the checklists.
- The peer-based inspection model uses teams of practicing professionals who are qualified through a CAP inspector training program.
Eligibility Requirements
- The CAP accredits laboratories that perform testing on specimens from human beings or animals, using methodologies and clinical applications within the expertise of the program. Laboratories must be appropriately licensed to perform testing when required by law.
- Laboratories outside the US seeking CAP accreditation must enroll in CAP PT/EQA programs for a minimum of six months before requesting an accreditation application.
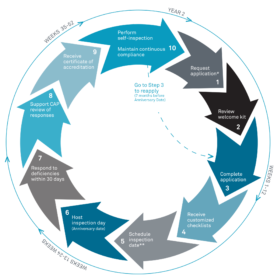
The Accreditation Cycle
The graphic below captures the accreditation and inspection cycle for CAP-accredited laboratories. For laboratories outside the US, subsequent inspections are announced.
Find a CAP-Accredited Laboratory
Use our database to search for CAP-accredited laboratories.