- Home
- Laboratory Improvement
- Reproductive Accreditation Program
Designed for the unique needs of reproductive laboratories in collaboration with the American Society for Reproductive Medicine, the CAP’s program promotes the quality and safety of laboratory services to give your physicians and patients confidence in the services it provides. Our program is recognized by the Society for Assisted Reproductive Technologies (SART) to meet membership requirements for in vitro fertilization facilities.
Annual updates of the Reproductive Laboratory Medicine checklist incorporate the latest best practices and advances in technology based on input from experts in the field.
Program Benefits
The Reproductive Accreditation Program:
- Focuses on areas to make laboratory processes more fail-safe and reduce risk of errors in patient identification, specimen labeling, handling of embryos and gametes and cryo storage conditions to protect your patients
- Includes the full spectrum of services that may be offered by a reproductive laboratory including embryology, andrology, endocrinology, cryopreservation, reproductive cell/tissue storage and preimplantation genetic screening/diagnosis
- Utilizes peer inspections from practicing embryologists to facilitate exchange of ideas and best practices to improve performance
- Includes requirements to evaluate the ongoing competency of all testing personnel and embryologists
- Gives your laboratory confidence in its preparation for FDA inspections for services involving the collection, processing, storage, or transplantation of donor tissues and cells
Program Details
- On-site inspections occur every two years using CAP Accreditation Checklists to assess compliance with program requirements.
- Participating laboratories can access the checklists through e-LAB Solutions Suite and non-accredited laboratories may purchase the checklist.
- Peer-based inspection model using practicing professionals as inspectors, who are qualified through a CAP inspector training program.
- For international laboratories seeking CAP accreditation, enrollment is required for a minimum of six months in our EQA/PT prior to requesting an accreditation application.
- CMS-approved accrediting organization for andrology and other tests regulated by CLIA ’88 and is recognized by The Joint Commission.
Eligibility Requirements
Laboratories that perform at least one embryology-related procedure or perform semen analysis and at least one additional high complexity test are eligible for this program.
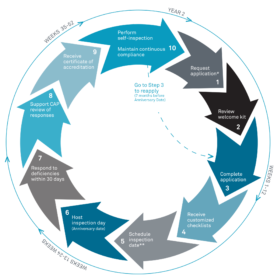
The Accreditation Cycle
The graphic below captures the accreditation and inspection cycle for CAP-accredited reproductive programs. Inspections for the Reproductive Laboratory accreditation program are announced.
Find a CAP-Accredited Reproductive Laboratory
Use our database to search for CAP accredited reproductive laboratories.