- Home
- Member Resources
- Articles
- The Expanding Role of Cell-Free DNA in Transfusion Medicine
Cell-free DNA (cfDNA) refers to short DNA fragments that circulate freely in the bloodstream, primarily originating from apoptotic and necrotic cells.1 In healthy individuals, most cfDNA is derived from hematopoietic cells; however, cfDNA can also originate from other sources, including fetal cells during pregnancy, donor tissues following organ or stem cell transplantation, pathogens, or malignant cells in cancer.
Initially identified in the 1940s, cfDNA garnered significant clinical attention in the late 1990s with the discovery of fetal cfDNA in maternal plasma, leading to the development of noninvasive prenatal screening for chromosomal disorders.2 Since then, cfDNA has revolutionized fields such as oncology, prenatal medicine, and solid organ transplantation. cfDNA is now emerging as a promising diagnostic and monitoring tool in transfusion medicine with applications that span prenatal care, transplant surveillance, microchimerism risk assessment, and potentially transfusion safety.
Clinical Applications of cfDNA in Transfusion Medicine
Noninvasive Fetal Blood Group Genotyping
One of the most clinically established uses of cfDNA in transfusion medicine is the noninvasive determination of fetal blood group antigens. cfDNA in the maternal plasma can be analyzed to determine the fetal RhD status and other clinically significant blood group antigens, such as Kell, Duffy, and RhC, without the need for invasive procedures.3
Prenatal identification of fetal RhD antigen status helps in the assessment of risk of RhD alloimmunization and in guiding the management of pregnancies at risk for hemolytic disease of the fetus and newborn.4 This application of cfDNA testing reduces unnecessary administration of Rh immunoglobulin (RhIG) to Rh-negative mothers, improving both safety and resource utilization. A comprehensive meta-analysis of 84 studies involving over 77,000 pregnancies confirmed that cfDNA-based genotyping for multiple fetal blood group antigens offers 99% to 100% sensitivity and specificity.3 Additionally, a 2025 clinical validation study utilized a commercially available, next-generation sequencing-based cfDNA test for fetal RhD status, aligning with updated ACOG recommendations that recognize cfDNA testing as an accepted methodology in this context.5 This study followed 665 RhD-negative pregnant patients, with 356 of 356 fetuses correctly identified as fetal RhD positive (sensitivity 100%, 95% CI, 98.9% to 100%), and 295 of 297 RhD-negative fetuses correctly identified as RhD negative (specificity 99.3%, 95% CI, 97.6% to 99.8%). The study utilized a large US cohort, confirming the feasibility of cfDNA testing within our diverse population. Implementing cfDNA testing could conserve the US RhIG supply through avoidance of unnecessary RhIG in up to 40% of RhD-negative pregnant patients. In several other parts of the world, cfDNA has already been implemented in clinical practice to guide antenatal RhIG prophylaxis.6 This approach minimizes the need for invasive procedures like amniocentesis and reduces the risk of alloimmunization by allowing targeted management during pregnancy.
Donor-Derived cfDNA for Transplant Monitoring and Apheresis Management
Donor-derived cell-free DNA (dd-cfDNA) is a powerful, minimally invasive biomarker for detecting allograft injury and acute rejection after solid organ transplantation (Figure 1).7 dd-cfDNA for transplant surveillance has been previously reviewed and will therefore not be covered here.8
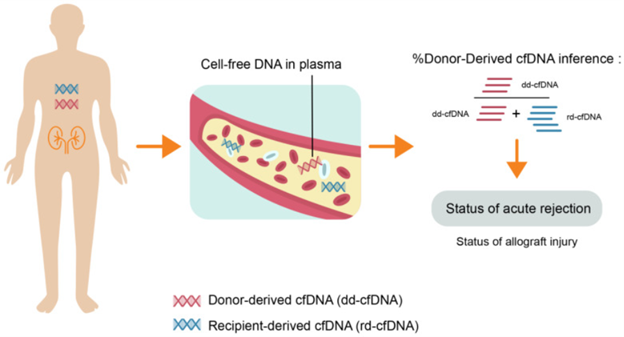
Transfusion medicine specialists are frequently consulted to initiate therapeutic plasma exchange (TPE) for antibody-mediated rejection (AMR), to reduce circulating donor-specific antibodies (DSAs). Currently, the Banff criteria (2022) utilize HLA DSA positivity for the diagnosis of active AMR; however, combining dd-cfDNA levels with DSA results can improve the positive predictive value for identifying patients with AMR.9 dd-cfDNA monitoring can also help assess treatment response, as declining levels suggest efficacy, while persistent elevations may signal ongoing allograft injury, guiding additional intervention. This allows transfusion medicine specialists to optimize TPE protocols, improve resource utilization, and enhance patient safety.
Potential Future Applications
Biomarker for Hematopoietic Activity and Cytopenias
cfDNA offers a novel, minimally invasive approach to evaluating hematopoiesis without the requirement of a bone marrow biopsy. cfDNA has been shown to correlate with cellular turnover, primarily from megakaryocytes (approximately 26%) and erythroblasts (1% to 4%).10 More studies are needed to determine whether cfDNA levels can serve as surrogate markers of platelet and red blood cell production, increasing in conditions such as thalassemia and decreasing during bone marrow suppression. Integration of cfDNA profiling with peripheral blood smear evaluation and bone marrow biopsy may enhance identification of the etiology of thrombocytopenia and anemia, thereby improving transfusion strategies and guiding targeted treatment decisions.
Microchimerism Surveillance
Microchimerism is the presence of allogeneic hematopoietic cells in an individual post-transfusion. Although investigational, cfDNA analysis can be used to detect microchimerism in transfused patients.11 Microchimerism is a common finding among frequently transfused patients, occurring in up to 79.7% to 81.2% of beta-thalassemia patients, with 68.7% demonstrating long-term microchimerism.11,12 The potential clinical consequences of microchimerism remain unclear, but immunosuppressive states or immune tolerance attributed to the disease itself and to repeated transfusions might at least in part explain this phenomenon. More studies are needed to determine the clinical significance of microchimerism, including whether detection by cfDNA alters clinical management or enables targeted intervention.
Pathogen Detection in the Transfusion Setting
Pathogen-derived cfDNA has emerged as valuable biomarkers in infectious diseases, enabling rapid and accurate diagnosis, treatment monitoring, and prognostic assessment.13,14 A commercial microbial cfDNA (mcfDNA) sequencing assay developed by Karius, detects and quantifies mcfDNA in plasma and can be utilized for diagnosing bacterial, viral, and fungal infections.15 This test works for sepsis and bloodstream infections, and it also detects localized infections like pneumonia and brain infections, as microbial DNA from any site releases into the bloodstream, making it a versatile tool for deep, hard-to-diagnose infections in various body parts. Similar approaches could be extended to blood donor screening. Currently, pathogen detection in blood products relies on targeted methods such as serological assays, nucleic acid testing, and immunoassays. In contrast, mcfDNA analysis offers the potential to serve as a more comprehensive blood donor screening tool capable of detecting multiple, emerging, or uncommon pathogens simultaneously. Furthermore, it could enhance surveillance for transfusion-transmitted infections, particularly in complex patient populations.
Conclusion
cfDNA is a promising biomarker that is reshaping transfusion medicine. Its proven utility in fetal blood group genotyping and transplant surveillance is expanding to encompass AMR monitoring, hematopoietic activity monitoring, and pathogen detection. As cfDNA assay technology improves and tissue-specific markers become standardized, the future of cfDNA in transfusion medicine will likely include personalized monitoring, improved resource use, and earlier detection of transfusion-related complications.
References
- Olson DR. The ‘liquid’ biopsy. College of American Pathologists. Accessed January 15, 2026. https://www.cap.org/member-resources/articles/the-liquid-biopsy
- Lo YM, Corbetta N, Chamberlain PF, et al. Presence of fetal DNA in maternal plasma and serum. Lancet. 1997;350(9076):485–487.
- Mustafa HJ, Najjariasl P, Aghajani et al. Diagnostic accuracy of cell-free DNA for the determination of fetal red blood cell antigen genotype: a systematic review and meta-analysis. Am J Obstet Gynecol. 2025; 233, 428-445.e16
- Committee on Practice Bulletins—Obstetrics. Practice Bulletin No. 181: Prevention of Rh D Alloimmunization. Obstet Gynecol. 2017;130(2):e57-e70.
- Gilstrop Thompson M, Xu W, Moore B, et al. Clinical Validation of a Prenatal Cell-Free DNA Screening Test for Fetal RHD in a Large U.S. Cohort. Obstet Gynecol. 2025;145(2):211-216.
- Clausen FB, Christiansen M, Steffensen R, et al. Report of the first nationally implemented clinical routine screening for fetal RHD in D- pregnant women to ascertain the requirement for antenatal RhD prophylaxis. Transfusion. 2012;52(4):752-758.
- Thongprayoon C, Vaitla P, Craici IM, et al. The Use of Donor-Derived Cell-Free DNA for Assessment of Allograft Rejection and Injury Status. J Clin Med. 2020 May 14;9(5):1480.
- Szczepanski J, Kim AS. Utilizing Cell-Free DNA Technologies for Clinically Significant Biomarkers in Solid Organ Transplantation. College of American Pathologists. Accessed February 18, 2026. https://www.cap.org/member-resources/articles/utilizing-cell-free-dna-technologies-for-clinically-significant-biomarkers-in-solid-organ-transplantation
- Jordan SC, Bunnapradist S, Bromberg JS, et al. Donor-derived Cell-free DNA Identifies Antibody-mediated Rejection in Donor Specific Antibody Positive Kidney Transplant Recipients. Transplant Direct. 2018 Aug 20;4(9):e379.
- Moss J, Ben-Ami R, Shai E, et al. Megakaryocyte- and erythroblast-specific cell-free DNA patterns in plasma and platelets reflect thrombopoiesis and erythropoiesis levels. Nat Commun. 2023 Nov 20;14(1):7542.
- Matsagos S, Verigou E, Kourakli A, et al. High Frequency of Post-Transfusion Microchimerism Among Multi-Transfused Beta-Thalassemic Patients. Front Med (Lausanne). 2022 Feb 16;9:845490.
- Matsagos S, Kourakli A, Argyropoulo, C, et al. High Frequency of Both, Short and Long-Term Post-Transfusion Microchimerism Among the Regularly-Transfused Thalassemic Patient Population. Blood. 2017;130(S1):3740.
- Hu Y, Zhao Y, Zhang Y, Chen W, Zhang H, Jin X. Cell-free DNA: a promising biomarker in infectious diseases. Trends Microbiol. 2025;33(4):421-433.
- Allos H, John TM, Stewart AG. Microbial cell-free DNA for diagnosis of bacterial and fungal infection in the immunocompromised host - what do we know?. Curr Opin Infect Dis. 2025;38(6):535-543.
- Park SY, Chang EJ, Ledeboer N, et al. Plasma Microbial Cell-Free DNA Sequencing from over 15,000 Patients Identified a Broad Spectrum of Pathogens. J Clin Microbiol. 2023;61(8):e0185522.

Pearl Audon, DO, is a 2nd year pathology resident at the University of California, Irvine. She received her doctorate from Western University of Health Sciences-College of Osteopathic Medicine in Pomona, CA and undergraduate from Scripps College in Claremont, CA. Passionate about increasing exposure to pathology, she started a summer laboratory internship program for high school and college students during her gap years and served as vice president of her medical school’s pathology interest group. Dr. Audon would like to be a laboratory director one day and has her eyes set on fellowships in transfusion medicine, translational research, and women’s pathology (breast and gynecology).

Gagan Mathur, MD, MBA, CPE, FCAP, is a board-certified transfusion medicine/blood banking physician and serves as the medical director of transfusion medicine at the University of California, Irvine (UCI). Dr. Mathur holds the academic title of associate professor of clinical pathology in the Department of Pathology & Laboratory Medicine at the UCI School of Medicine. He pursued his master of business administration (MBA) from the University of Iowa Tippie School of Management, along with his clinical training in clinical pathology at the University of Iowa Hospitals & Clinics. Dr. Mathur earned the title of Certified Physician Executive from American Association for Physician Leadership. Dr. Mathur’s areas of clinical interest and expertise include patient blood management, therapeutic apheresis, and cellular therapies. Along with his clinical responsibilities, Dr. Mathur is enthusiastically involved in clinical research and teaching to accomplish his academic commissions. Utilizing his interest and training in business management, informatics, and healthcare administration, Dr. Mathur strives to provide effective and efficient patient care.